

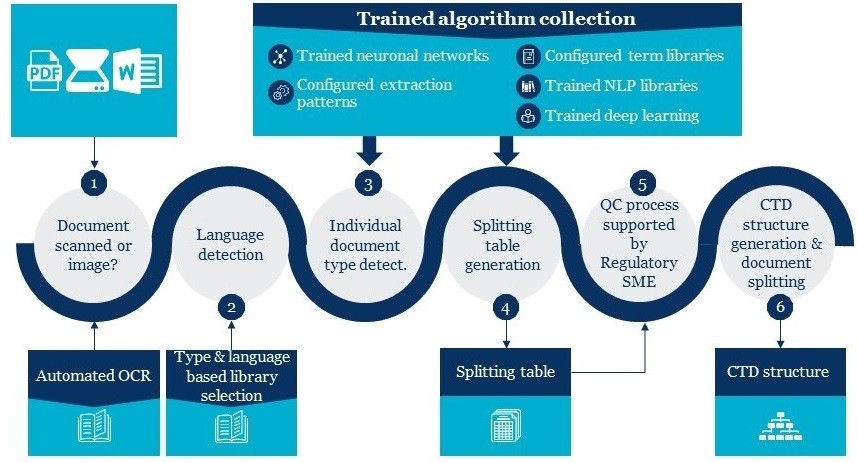
Cyclone Pharma has experts for eCTD Compilation and conversion servcies,CPPL had developed first eCTD software when eCTD Draft guidelines have been declared for Industry.Cyclone team have deep knowledge and understanding of eCTD Compilation and conversion.
What we offer is a comprehensive range of services to
assist in regulatory compliance. Our team at Cyclone Pharmaceuticals Pvt Ltd, a leading
pharmaceutical consultancy in India, specializes in writing dossiers and converting them
into eCTD format. We also handle the submission process to the appropriate regulatory
authorities. Our eCTD conversion software, developed by our expert consultants at CPPL,
simplifies the process and significantly reduces submission costs compared to market
standards.
For all your regulatory compilations, document creation, and submission needs, our team
at CPPL is here to assist you at every stage. Unlike other consultants, we have a dedicated
team with expertise in QA, QC, Production, and Regulatory sectors, ensuring thorough
document review, dossier compilation, and prompt query responses.
Our regulatory consultancy services at Cyclone Pharmaceuticals have a proven track
record of delivering high-quality solutions at the lowest cost. Many times, our compiled
documents have been accepted by regulatory authorities without any queries. We are always
updated with the latest regulations and can provide accurate compilation in various formats,
including Drug Dossier, DMF, CTD, ACTD, KDMF, USDMF, eCTD, EUDMF, USDMF, and more.
Regulatory consultancy services of Cyclone Pharmaceuticals are proven services with
highest Quality and Lowest cost.
Our compiled documents many times are being accepted by regulatory authorities without
single query. CPPL team is always
alert for new updations in regulations provides perfect Drug Dossier, DMF, CTD, ACTD, KDMF,
USDMF. eCTD, EUDMF, USDMF and
other format compilation.